septembre 2021
Publications les plus recentes
Airagnes G, du Vaure CB, Galam E, Bunge L, Hoertel N, Limosin F, Jaury P, Lemogne C. Personality traits are associated with cognitive empathy in medical students but not with its evolution and interventions to improve it. J Psychosom Res. 2021 May;144:110410. doi: 10.1016/j.jpsychores.2021.110410. Epub 2021 Feb 26. PMID: 33676151 Clinical Trial. Download from HAL-INSERM
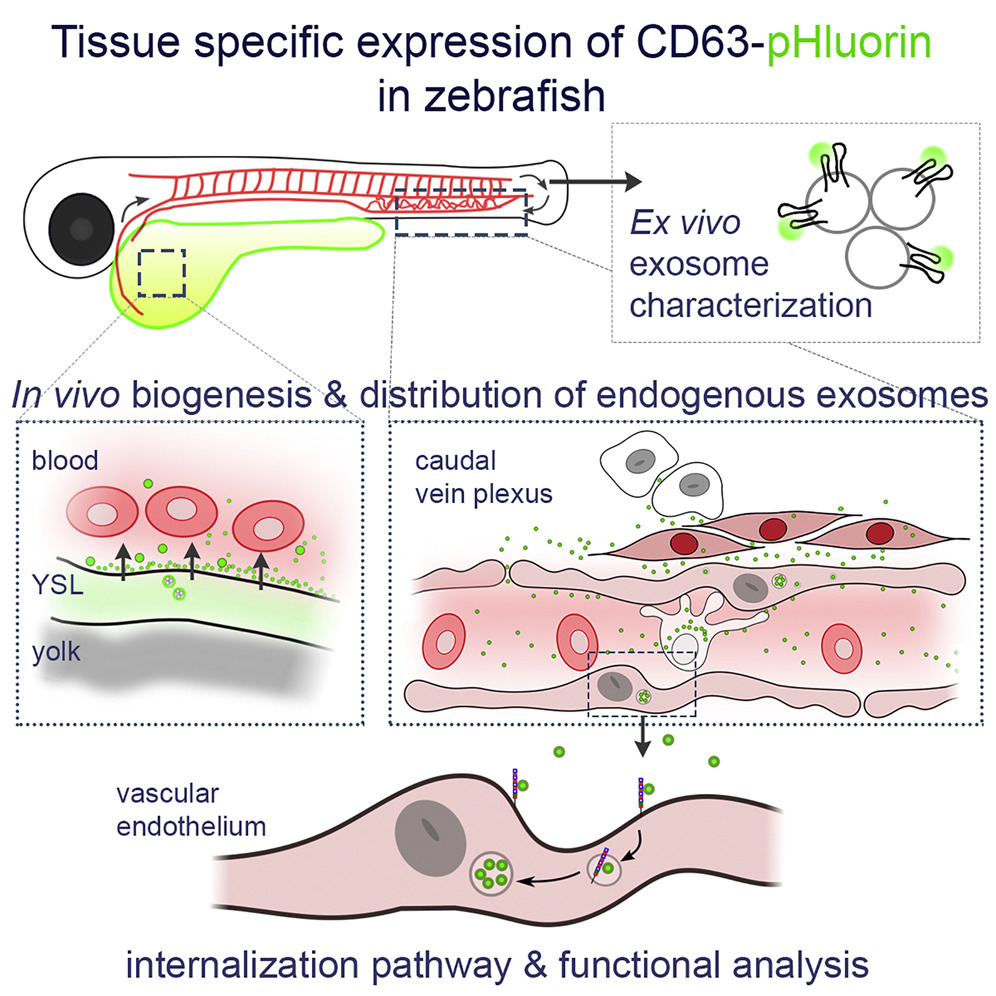
Androuin A, Verweij FJ, van Niel G. Zebrafish as a preclinical model for
Extracellular Vesicle-based therapeutic development.
Adv Drug Deliv Rev. 2021 May 28:S0169-409X(21)00189-7. doi:
10.1016/j.addr.2021.05.025. Online ahead of print. Review. PubMed
PMID: 34058284.
Arnaud C, Boulanger M, Lorthioir A, Amar L, Azarine A, Boyer L,
Chatellier G, Di Monaco S, Jeunemaitre X, Kastler A, Mousseaux E,
Oppenheim C, Thony F, Persu A, Olin JW, Azizi M, Touzé E. Male Sex Is Associated With Cervical Artery
Dissection in Patients With Fibromuscular Dysplasia. J
Am Heart Assoc. 2021 May 17;:e018311. doi: 10.1161/JAHA.120.018311.
[Epub ahead of print] PubMed PMID: 33998257. OPEN ACCES. Download
from HAL-INSERM
Baron JC. Nerinetide: A Potential
Neuroprotectant as Adjunct to Thrombectomy for Acute
Stroke. Can J Neurol Sci. 2021 Jan;48(1):138. doi:
10.1017/cjn.2020.183. Epub 2020 Aug 20. PMID: 32814602 No abstract
available Download from HAL-INSERM
Baron JC. The core/penumbra model:
implications for acute stroke treatment and patient selection in
2021. Eur J Neurol. 2021 Aug;28(8):2794-2803. doi:
10.1111/ene.14916. Epub 2021 Jun 18.PMID: 33991152 Review.
Ben Hassen W, Touloupas C, Benzakoun J, Boulouis G, Bretzner M, Bricout N, Legrand L, Rodriguez C, Le Berre A, Seners P, Turc G, Cordonnier C, Oppenheim C, Henon H, Naggara O. Impact of Repeated Clot Retrieval Attempts on Infarct Growth and Outcome After Ischemic Stroke. Neurology. 2021 Jun 23;. doi: 10.1212/WNL.0000000000012321. [Epub ahead of print] PubMed PMID: 34162721.
Benzakoun J, Charron S, Turc G, Hassen WB, Legrand L, Boulouis G,
Naggara O, Baron JC, Thirion B, Oppenheim C. Tissue outcome prediction in hyperacute
ischemic stroke: Comparison of machine learning
models. J Cereb Blood Flow Metab. 2021 Jun
23:271678X211024371. doi: 10.1177/0271678X211024371. Online ahead
of print. PubMed PMID: 34159824.
Berendsen S, Kapitein P, Schirmbeck F, van Tricht MJ, McGuire P, Morgan C, Gayer-Anderson C, Kempton MJ, Valmaggia L, Quattrone D, di Forti M, van der Gaag M, Kirkbride JB, Jongsma HE, Jones PB, Parellada M, Arango C, Arrojo M, Bernardo M, Sanjuán J, Santos JL, Szöke A, Tortelli A, Llorca PM, Tarricone I, Tripoli G, Ferraro L, La Cascia C, Lasalvia A, Tosato S, Menezes PR, Del-Ben CM, Nelson B, Riecher-Rössler A, Bressan R, Barrantes-Vidal N, Krebs MO, Nordentoft M, Ruhrmann S, Sachs G, Rutten BPF, van Os J, Velthorst E, de Haan L. Pre-training inter-rater reliability of clinical instruments in an international psychosis research project. Schizophr Res. 2021 Apr;230:104-107. doi: 10.1016/j.schres.2020.08.001. Epub 2020 Nov 24. PubMed PMID: 33243716.
Berge E, Whiteley W, Audebert H,
Marchis GM, Fonseca AC, Padiglioni C, de la Ossa NP, Strbian D,
Tsivgoulis G, Turc G. European Stroke Organisation (ESO)
guidelines on intravenous thrombolysis for acute ischaemic
stroke. Eur Stroke J. 2021 Mar;6(1):I-LXII. doi:
10.1177/2396987321989865. Epub 2021 Feb 19. PubMed PMID: 33817340;
PubMed Central PMCID: PMC7995316. OPEN ACCES. Download from
HAL-INSERM
Bergeret S, Queneau M, Rodallec M, Curis E, Dumurgier J, Hugon J,
Paquet C, Farid K, Baron JC. [18 F]FDG PET may differentiate cerebral amyloid
angiopathy from Alzheimer disease. Eur J Neurol. 2021
May;28(5):1511-1519. doi: 10.1111/ene.14743. Epub 2021 Feb 3.PMID:
33460498