Team leader : Guillaume Turc
Team member : Anaelle Alouit | Caroline Arquizan | Paul-Peter Arslan | Jean-Claude Baron | Sarah Benghanem | Eléonore Bouchereau | David Calvet | Nicolas Chausson | Florence Colle | Camille Des Laurriers | Lucile Dupin | Elisa Dziezuk | Martine Gavaret | Sonia Hamdoun | Bertrand Hermann | Clothilde Isabel | Manoëlle Kossorotoff | Quentin Le Boterff | Pavel Lindberg | Anaïs Llorens | Jean-Louis Mas | Estelle Pruvost-Robieux | Pierre Seners | Tarek Sharshar | Didier Smadja | Maxime Teremetz
 |
Stroke is a very common disorder and a major cause of death, disability and dementia worldwide. The aims of our research are to: (i) identify/better define determinants of stroke prognosis, in terms of response to acute treatment, risk of recurrent stroke and other vascular events, and long-term consequences of brain injury, such as functional and motor outcome, post-stroke cognitive decline or depression), and (ii) assess the benefit–risk ratio of therapeutic strategies through randomized clinical trials. We endeavour to answer clinical questions with direct applications in clinical practice. In addition to clinical expertise in stroke allowing for accurate phenotyping (stroke being a highly heterogeneous disorder), our team has expertise in methodology of clinical research and biostatistics. We have also recently engaged into proof-of-principle, mechanistic and translational studies.
|
Summary
Ischemic stroke results from a focal reduction in cerebral blood flow (CBF) due to the sudden occlusion of a brain artery caused by a blood clot. In areas with CBF below a specific ‘penumbral’ threshold the neurons stop firing, causing clinical symptoms, but are still viable and can be rescued by early reperfusion1. Penumbral salvage by early reperfusion results in proportional clinical recovery. Otherwise, reduced oxygen supply triggers a complex pathophysiological cascade ultimately leading to tissue infarction, such that the region of irreversible cell death, the so-called ‘core’, grows over time at the expense of the viable penumbra (Figure).
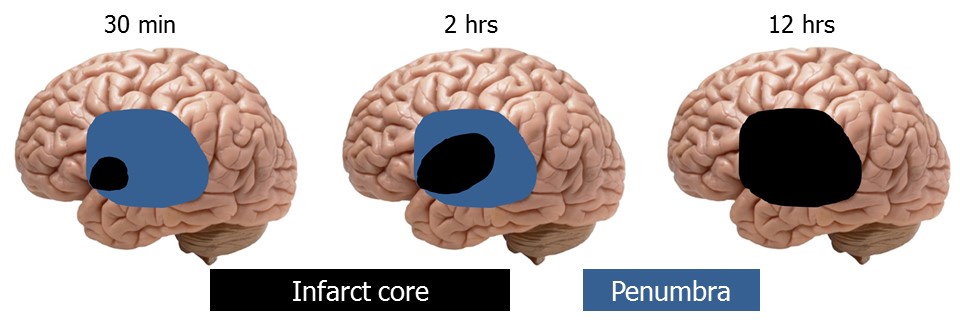
Reperfusing as much penumbra as early as possible by intravenous thrombolysis (IVT) and/or endovascular thrombectomy is therefore the goal of stroke management. However, reperfusion therapies do not allow timely recanalization of the occluded artery in all patients to save the entire penumbra, resulting in still unsatisfactory outcome in the majority of cases. In addition, some patients have early (within 24 hrs) neurological deterioration (END) after IVT, which can be due to various causes, particularly hemorrhagic transformation (HT) or remain unexplained. Thus, how best to select and triage candidates for such therapies remains unclear, while the mechanisms underlying HT and unexplained END are still largely unknown. In order to address these issues, we established a prospective clinical, biological and radiological database of acute stroke patients, candidates for reperfusion therapies. We have shown that unexplained END is related to extra-penumbral infarct growth, notably secondary to extension of the original thrombus2,3. Regarding HT, we have shown that a high burden of cerebral microbleeds (i.e., asymptomatic micro-hemorrhages reflecting underlying chronic small vessel disease) is independently associated with both symptomatic intracranial hemorrhage and poor 3-month outcome4,5, suggesting that thrombectomy might be a better option than IVT in such patients. To improve acute stroke management, we have developed a score to predict 3-month outcome after IVT, based on data readily available at patient admission6. We have also assessed the value of 13 clinical scales to predict large artery occlusion and found that using them for prehospital triage would result in a loss of opportunity for ≥20% of patients who would be inappropriately sent to a center lacking neuro-interventional facilities7. We therefore suggest that intracranial arterial imaging be performed in all stroke patients presenting within 6hrs.
A promising novel approach to enlarge the volume of salvageable penumbra at reperfusion time, and in turn improve final outcome, is to ‘freeze’, i.e. prevent further demise of, the penumbra until reperfusion occurs. Given that the penumbra is severely hypoxic, normobaric oxygen therapy (NBO) is an appealing potential approach to freeze the penumbra. We have shown in a rodent stroke model with early reperfusion that NBO almost entirely prevents both neuronal death and sensorimotor deficits8, suggesting this straightforward approach might be a valuable adjunct to reperfusion therapies, applicable from the pre-hospital stage.
Our goal for the next 5 years is to optimize the management of, and improve functional outcome from, acute stroke. To this end, we aim to better understand and address the mechanisms underlying early recanalization, the demise of the ischemic penumbra and functional outcome. We will (i) develop additional therapeutic approaches through translational research to freeze the penumbra; (ii) evaluate new strategies of acute stroke management by RCTs; and (iii) pursue our research on determinants of outcome following acute stroke.
Although recanalisation therapies salvage the ischemic penumbra and in doing so effectively improve clinical outcome, they can only rescue that amount of penumbra still present at reperfusion time. As mentioned above, a new and promising approach to enlarge the amount of rescued penumbra and hence further improve outcome is to ‘freeze’ the penumbra until reperfusion occurs. Based on pre-clinical evidence, the penumbral ‘freezing’ concept will be tested in two pilot RCTs, one testing whether cathodal transcranial direct cortical stimulation (tDCS) - shown in rodents to reduce the occurrence of peri-infarct depolarizations and thus infarction of the penumbra - prevents early infarct growth; and the other, to be tested in 3 centres (Paris, Berlin, New-York; funded by Leducq Foundation), whether somatosensory stimulation can protect the penumbra and constrain growth of the diffusion lesion. Based on our findings in rodent models (see above), our team will also nationally coordinate the EU-funded PROOF study, a phase II proof-of-concept trial of penumbral rescue by normobaric oxygen therapy in stroke patients harboring a sizeable penumbral tissue volume.
The recent demonstration of the efficiency of bridging therapy (IVT followed by mechanical thrombectomy), represents a major breakthrough but also challenges the care system, notably in the prehospital setting. Indeed, thrombectomy is only indicated in selected patients with large artery occlusion (LAO), and its beneficial effects are highly time-dependent. Moreover, few stroke centres have an on-site, 24/7 neurointerventional team able to perform thrombectomy. Therefore, patient selection through multimodal imaging is essential. It is usually performed in primary stroke centres, which implies that those patients fulfilling the criteria for thrombectomy are immediately transferred to a comprehensive stroke centre. This process leads to important delays and, hence, potentially worse outcomes. To improve early patient management, we have designed the ASPHALT study, a multicenter RCT evaluating the impact on 3-month outcome and cost-efficiency of a mobile stroke unit (dedicated ambulance equipped with a CT scanner and telemedicine) for acute stroke management and triaging. This should not only allow starting IVT earlier, i.e., on the field, but could also help identify candidates for additional thrombectomy and take them directly to the appropriate stroke centre. It also provides unparalleled opportunity to study serum biomarkers in confirmed ischemic strokes seen at very early time points, which might in turn lead to new approaches for prehospital patient triaging. Global collaboration on prehospital stroke studies is planned through the PRESTO network.
To address the issue of how best to select, among patients thrombolyzed in primary stroke centers, those to be transferred to comprehensive stroke centres for thrombectomy, we are currently running a multi-centre study to determine the best predictors of no early recanalization. The occurrence of early recanalization will be determined on transcranial Doppler over 2hrs after start of IVT or on the pre-thrombectomy angiography; all patients will have MR-based assessment of core and penumbra, collateral status and thrombus characteristics, and blood samples to assess biomarkers of thrombus resistance, including NETs. In addition to early recanalization, this study will provide novel insights into the pathophysiology of thrombus resistance in acute stroke. Finally, we will pursue our research towards developing predictive models of the early and long-term clinical evolution after reperfusion therapies, and in turn inform clinical decision-making and future RCTs.
1. CAROTID STENOSIS DUE TO ATHEROSCLEROSIS
Symptomatic carotid stenosis
Carotid artery stenosis due to atherosclerosis is responsible for up to 15% of all ischemic strokes and transient ischemic attacks (TIAs). Patients presenting with a first ischemic cerebral event due to atherosclerotic carotid stenosis (so-called symptomatic carotid stenosis) have a high risk of stroke recurrence. Surgically removing the carotid plaque by carotid endarterectomy (CEA) reduces the risk of stroke recurrence but carries a non-negligible risk of perioperative stroke or death. Carotid angioplasty and stenting (CAS) has been introduced as a less invasive alternative to surgery. However, whether stenting is as safe as surgery and provides similar long-term protection against stroke has been a subject of considerable debate. To answer these questions, we conducted the EVA-3S study - a multicentre, RCT funded by the French Department of Health - to compare CAS to CEA in patients with symptomatic carotid artery stenosis. The main results of this trial are that CAS (i) carries a higher risk of procedural stroke than CEA, but (ii) is as effective as CEA to prevent ipsilateral stroke recurrence up to 10 years following the intervention9-13. To estimate size effects more robustly and investigate whether an intervention is more or less effective for different types of participants using meta-analyses of individual patient data, we founded the Carotid Stenosis Trialists’ Collaboration, a large international database from all the European and US trials comparing CAS to CEA. In the last few years, we have identified several factors associated with a higher procedural risk of CAS, such as anatomical factors, operator experience, technical aspects and timing of revascularisation14-16. In particular, we recently reported that age has a strong effect on the procedural risk of CAS, but little effect on the procedural risk of CEA. The procedural risk of stroke with CAS is twice that with CEA in patients 70 yrs or older, whereas it is similar in patients <70yrs. We also showed that age had little effect on the long-term durability (postprocedural stroke risk) for either CAS or CEA14. These findings are now used in international guidelines to select the appropriate intervention according to individual patient’s characteristics.
Asymptomatic carotid stenosis
One approach to prevent carotid stroke is to remove the plaque before stroke occurs, that is, so-called asymptomatic carotid stenosis, the incidence of which is ~2.5% in the general population. Despite early RCTs supporting a role for CEA in the management asymptomatic carotid disease, there is little international consensus regarding how best to treat this condition in the modern era. This is first because the benefit of CEA in RCTs was marginal (absolute risk reduction of ipsilateral stroke: 1%/yr, i.e., from 2%/yr with medical treatment down to 1%/yr with CEA), and second because recent advances in medical therapy currently confer a risk of stroke < 1%/yr. It is therefore uncertain whether the procedural risk of stroke or death related to CEA is still justified. In parallel, imaging strategies based mainly on ultrasound and MRI have emerged to identify unstable plaques (i.e., with recent inflammatory activity favoring thrombus formation and emboli) and those patients with carotid stenosis at higher risk of ipsilateral stroke. Our hypothesis is that the use of these predictors can identify a subset of such patients who would benefit from CEA. Over the next 5 years, we will test this hypothesis in ACTRIS, a multicenter RCT funded by the French Department of Health (NCT0056; to start in 2017) comparing CEA + optimal medical therapy to optimal medical therapy alone, in patients with asymptomatic carotid stenosis at higher-than-average risk of ipsilateral stroke, based on currently identified predictors including intraplaque hemorrhage on MRI, TCD-detected micro-embolic signals, rapid progression of stenosis and impaired cerebrovascular reserve. We will also investigate whether new imaging or serum biomarkers of plaque instability reliably predict the occurrence of ipsilateral stroke. In this respect, we have recently completed a study to determine to what extent plasma levels of Lp-PLA2 (a potential biomarker of unstable plaques) relate to local plaque inflammation and composition (as assessed by FDG-PET imaging, high-resolution MRI and histological examination) and to assess the value of Lp-PLA2 to identify unstable carotid plaques. We will also co-coordinate a workpackage of CVENT (H2020 programme), which will assess the risk of plaque rupture by multi-spectral photoacoustic imaging (to start in late 2017).
2. PATENT FORAMEN OVALE (PFO) AND ATRIAL SEPTAL ANEURYSM (ASIA)
The cause of ischemic stroke remains unknown (or “cryptogenic”) in up to 40% of cases. The foramen ovale is a natural interatrial channel, which normally closes after birth. In about 25% of people, the foramen remains patent throughout life, maintaining a channel through which a venous thrombus may reach the systemic circulation and cause stroke (‘paradoxical embolism’). The relationship between persistence of a PFO and ischaemic stroke has long been one of our main topics of interest. Our major contributions to this field were to show that young adults with a cryptogenic ischemic stroke more often have a PFO than control subjects, to quantify the risk of stroke recurrence and to identify a subgroup of patients with both a PFO and an atrial septum aneurysm (ASA) that carriers a high risk of stroke recurrence17. However, many uncertainties persist with regard to the causative relationship between PFO and stroke in the individual patient, stroke mechanisms, and the best strategy to prevent stroke recurrence18,19. As PFO is common in the general population, it may coexist by chance alone in patients with cryptogenic stroke. To resolve this issue, we contributed to develop a model that can estimate the patient-specific probability that a PFO is pathogenically related to the index stroke, and identify those patients most likely to benefit from PFO closure20. With regard to stroke prevention, we have conducted the CLOSE study - an academic, multicentre (32 French and German centres) RCT, funded by the French Department of Health (NCT00562289) - to compare PFO closure, oral anticoagulants and antiplatelet therapy, for secondary stroke prevention in 665 cryptogenic stroke patients aged 16 to 60 years with a large PFO or a PFO + ASA21. The trial has just been completed (mean follow-up of patients, 5,5 yrs). The results will be presented at the next European Stroke Organisation Congress (Prague, May 2017) and are expected to have a major impact on current guidelines. Thanks to its design, CLOSE will also test the hypothesis that paradoxical embolism (or cardiogenic embolism) is a major stroke mechanism in these patients. Given the low rate of stroke recurrence in young patients with PFO-related stroke, a pooled analysis of individual patient data from all RCTs will be needed to identify patient subgroups with different responses to treatment.
3. CEREBRAL SMALL VESSEL DISEASE
Cerebral small vessel disease (SVD) associated with aging and hypertension is responsible for about 30 % of ischemic strokes and underlie up to 60-70% of intracerebral haemorrhages. It also plays a major role in age-related cognitive decline. However, the pathophysiology of SVD remains largely unknown. In the next 5 years, we plan to assess clinical, imaging and biological predictors of SVD progression. Specifically, we will assess the cross-talk between large artery disease and cerebral SVD. Indeed, the lumen of proximal large arteries enlarges with aging and hypertension, and their arterial wall both thickens and stiffens. Evidence suggests that increased arterial pulsatility due to large artery stiffening is transmitted to cerebral small arteries and related to brain white-matter lesions. Our objectives are to assess (i) the association between large artery stiffness (determined from pulse-wave velocity) and severity of SVD (as assessed by MRI) and (ii) the impact of large artery stiffness on occurrence of SVD complications such as stroke recurrence, cognitive decline and disability. To this end, we will take advantage of the DHU Neurovasc clinical-radiological database and a biobank of 500 patients with lacunar infarcts due to SVD who will be followed for up to 5 years. This cohort, funded by the DHU, will start in early 2017. In addition, we plan a translational approach to assess mechanisms underlying brain damage in SVD. On one hand, we will take part of the RHU program (TRT_cSVD) of our DHU, whose objective is to take advantage of known genetic SVDs, such as CADASIL, to identify innovative biomarkers and clinical tools for therapeutic evaluation in patients. On the other hand, we aim to assess whether neuro-glio-vascular interactions could explain brain damage in common forms of SVD. Molecular and functional explorations of the neurovascular unit will be assessed by M Cohen-Salmon, (Collège de France) in order to unravel the molecular mechanisms involved in SVD, define new biological markers of the disease and develop innovative therapeutical strategies.
Up to 80% of stroke patients have impaired manual dexterity reducing their independence, return to work and quality of life. A key clinical and research challenge is to design interventions that optimize brain plasticity to improve recovery of dexterity. Intense mobilization or stimulation with transcranial direct current stimulation (tDCS) does not seem beneficial for improving recovery when applied very early after stroke. In later phases after stroke, existing therapy approaches do improve upper limb function in general, however, most show modest functional gains. We have therefore recently developed the Finger Force Manipulandum (FFM) which quantifies the forces applied by the fingers during various tasks (Fig.2). The FFM allows objective measurement of multiple dexterity components including independent finger movements, which are crucial for dexterous control yet absent in existing clinical assessments. A recent validation study has shown that this patented method is feasible and allows identification of individual dexterity profiles in stroke patients22. Our working hypothesis is that individually tailored dexterity training in stroke patients will improve hand function recovery, spontaneous use in the home-setting, and brain plasticity. We also hypothesize that combining specific dexterity training with suitable neuromodulation techniques (such as TMS, tDCS) will enhance brain plasticity and motor recovery even further. A proof-of-concept RCT study (funded by SATT, IDF-Innovation) will start in 2017 to compare targeted training of dexterity with FFM with conventional rehabilitation. We will also assess how lesion location relates to various components of dexterity using calculation of corticospinal tract lesion load (Figure) and voxel-based lesion symptom mapping. TMS and fMRI will be used to identify the neural correlates of successful dexterity recovery (especially independence of finger movements). Another study will evaluate the effect of cerebellar tDCS on motor learning and retention of finger movement sequences in stroke patients (partially funded by Gossweiler Foundation, applications submitted). Previous studies in healthy subjects have shown that cerebellar tDCS can enhance motor learning. However, studies in stroke patients are lacking. The rationale in our study here is that non-invasive anodal tDCS can reduce cerebellar inhibition of motor cortex and thus enhance learning of finger sequences. High resolution 128 channel EEG will be used to study electrical activity modulation in motor cortex during and post cerebellar tDCS.
Figure. Research methods
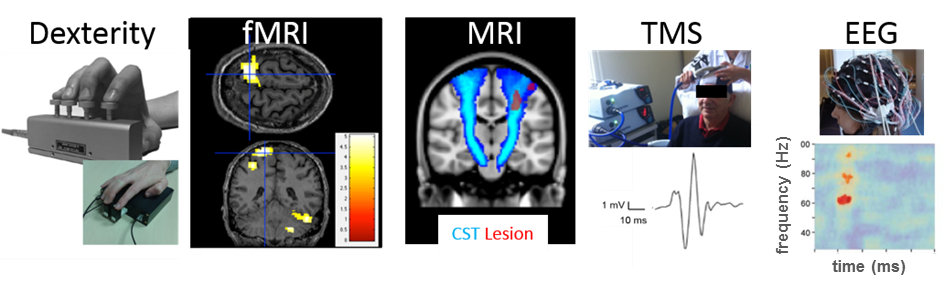
Dexterity is quantified using the patented Finger Force Manipulandum and using novel MRI compatible finger force device Pilot fMRI shows expected force-related brain activation in contralateral M1 (crosshairs) and ipsilateral cerebellum during task performance (FDR corrected, P<0.05). Structural and functional integrity of corticospinal tract is measured using anatomical MRI to calculate cortico-spinal tract lesion load and motor evoked potentials using TMS. EEG will be used to study time-frequency representations and corticomuscular coherence during dexterity tasks after cerebellar tDCS.
1 / Baron JC. Mapping the ischaemic penumbra with PET: implications for acute stroke treatment. Cerebrovasc. Dis., 1999,9:193-201.
2 / Tisserand M, Seners P, Turc G, Legrand L, Labeyrie MA, Charron S, Meder JF, Mas JL, Oppenheim C, Baron JC. Mechanisms of Unexplained Neurological Deterioration After Intravenous Thrombolysis. Stroke. 2014;45:3527-3534.
3 / Seners P, Hurford R, Tisserand M, Turc G, Legrand L, Naggara O, Mas JL, Oppenheim C, Baron JC. Is Unexplained Early Neurological Deterioration After Intravenous Thrombolysis Associated With Thrombus Extension? Stroke. 2016;(Published ahead of print).
4 / Turc G, Sallem A, Moulin S, Tisserand M, Machet A, Edjlali M, Baron JC, Leclerc X, Leys D, Mas JL, Cordonnier C, Oppenheim C. Microbleed Status and 3-Month Outcome After Intravenous Thrombolysis in 717 Patients With Acute Ischemic Stroke. Stroke. 2015;46:2458-2463.
5 / Tsivgoulis G, Zand R, Katsanos AH, Turc G, Nolte CH, Jung S, Cordonnier C, Fiebach JB, Scheitz JF, Klinger-Gratz PP, Oppenheim C, Goyal N, Safouris A, Mattle HP, Alexandrov AW, Schellinger PD, Alexandrov AV. Risk of Symptomatic Intracerebral Hemorrhage After Intravenous Thrombolysis in Patients With Acute Ischemic Stroke and High Cerebral Microbleed Burden: A Meta-analysis. JAMA Neurology. 2016;73:675-683.
6 / Turc G, Apoil M, Naggara O, Calvet D, Lamy C, Tataru AM, Meder JF, Mas JL, Baron JC, Oppenheim C, Touze E. Magnetic Resonance Imaging-DRAGON Score 3-Month Outcome Prediction After Intravenous Thrombolysis for Anterior Circulation Stroke. Stroke. 2013;44:1323-1328.
7 / Turc G, Maier B, Naggara O, Seners P, Isabel C, Tisserand M, Raynouard I, Edjlali M, Calvet D, Baron JC, Mas JL, Oppenheim C. Clinical Scales Do Not Reliably Identify Acute Ischemic Stroke Patients With Large-Artery Occlusion. Stroke. 2016;47:1466-U1210.
8 / Ejaz S, Emmrich JV, Sitnikov SL, Hong YT, Sawiak SJ, Fryer TD, Aigbirhio FI, Williamson DJ, Baron JC. Normobaric hyperoxia markedly reduces brain damage and sensorimotor deficits following brief focal ischaemia. Brain. 2016;139:751-764.
9 / Mas JL, Chatellier G, Beyssen B, et al; EVA-3S Investigators. Endarterectomy versus stenting in patients with symptomatic severe carotid stenosis. New England Journal of Medicine. 2006;355:1660-1671.
10 / Mas JL, Trinquart L, Leys D, et al ; EVA-3S Investigators. Endarterectomy Versus Angioplasty in Patients with Symptomatic Severe Carotid Stenosis (EVA-3S) trial: results up to 4 years from a randomised, multicentre trial. Lancet Neurology. 2008;7:885-892.
11 / Bonati LH, Dobson J, Algra A, Branchereau A, Chatellier G, Fraedrich G, Mali WP, Zeumer H, Brown MM*, Mas JL*, Ringleb PA*. Short-term outcome after stenting versus endarterectomy for symptomatic carotid stenosis: a preplanned meta-analysis of individual patient data. Lancet. 2010;376:1062-1073 (* Equal contribution)
12 / Arquizan C, Trinquart L, Touboul PJ, Long A, Feasson S, Terriat B, Gobin-Metteil MP, Guidolin B, Cohen S, Mas JL, for the EVA-3S Investigators. Restenosis Is More Frequent After Carotid Stenting Than After Endarterectomy The EVA-3S Study. Stroke. 2011;42:1015-1020.
13 / Mas JL, Arquizan C, Calvet D, Viguier A, Albucher JF, Piquet P, Garnier P, Viader F, Giroud M, Hosseini H, Hinzelin G, Favrole P, Henon H, Neau JP, Ducrocq X, Padovani R, Milandre L, Rouanet F, Wolff V, Saudeau D, Mahagne MH, Sablot D, Amarenco P, Larrue V, Beyssen B, Leys D, Moulin T, Lievre M, Chatellier G, for the EVA-3S Investigators. Long-Term Follow-Up Study of Endarterectomy Versus Angioplasty in Patients With Symptomatic Severe Carotid Stenosis Trial. Stroke. 2014;45:2750-2756
14 / Howard G, Roubin GS, Jansen O, Hendrikse J, Halliday A, Fraedrich G, Eckstein HH, Calvet D, Bulbulia R, Bonati LH, Becquemin JP, Algra A, Brown MM*, Ringleb PA*, Brott TG*, Mas JL*, for the Carotid Stenting Trialists Collaboration. Association between age and risk of stroke or death from carotid endarterectomy and carotid stenting: a meta-analysis of pooled patient data from four randomised trials. Lancet. 2016;387:1305-1311 (* Equal contribution)
15 / Naggara O, Touze E, Beyssen B, Trinquart L, Chatellier G, Meder JF, Mas JL, for the EVA 3S Investigators. Anatomical and Technical Factors Associated With Stroke or Death During Carotid Angioplasty and Stenting Results From the Endarterectomy Versus Angioplasty in Patients With Symptomatic Severe Carotid Stenosis (EVA-3S) Trial and Systematic Review. Stroke. 2011;42:380-388
16 / Calvet D, Mas JL, Algra A, Becquemin JP, Bonati LH, Dobson J, Fraedrich G, Jansen O, Mali WP, Ringleb PA, Chatellier G, Brown MM. Carotid Stenting: Is There an Operator Effect? A Pooled Analysis From the Carotid Stenting Trialists' Collaboration. Stroke. 2014;45:527-532
17 / Mas JL, Arquizan C, Lamy C, et al. Patent Foramen Ovale and Atrial Septal Aneurysm Study Group. Recurrent cerebrovascular events associated with patent foramen ovale, atrial septal aneurysm, or both. N Engl J Med 2001; 345: 1740–46.
18 / Ferro JM, Massaro AR, Mas JL. Aetiological diagnosis of ischaemic stroke in young adults. Lancet Neurology. 2010;9:1085-1096.
19 / Calvet D, Mas JL. Closure of patent foramen ovale in cryptogenic stroke: a never ending story. Current Opinion in Neurology. 2014;27:13-19
20 / Kent DM, Ruthazer R, Weimar C, Mas JL, Serena J, Homma S, Di Angelantonio E, Di Tullio MR, Lutz JS, Elkind MSV, Griffith J, Jaigobin C, Mattle HP, Michel P, Mono ML, Nedeltchev K, Papetti F, Thaler DE. An index to identify stroke-related vs incidental patent foramen ovale in cryptogenic stroke. Neurology. 2013;81:619-625.
21 / Mas JL, Derumeaux G, Amarenco P, et al; CLOSE Investigators. Closure of patent foramen ovale, oral anticoagulants or antiplatelet therapy to prevent stroke recurrence: Study design. International Journal of Stroke. 2016;11:724-732.
22 / Térémetz M, Colle F, Hamdoun S, Maier MA, Lindberg PG. A novel method for the quantification of key components of manual dexterity after stroke. J Neuroeng Rehabil. 2015 Aug 2;12:64.